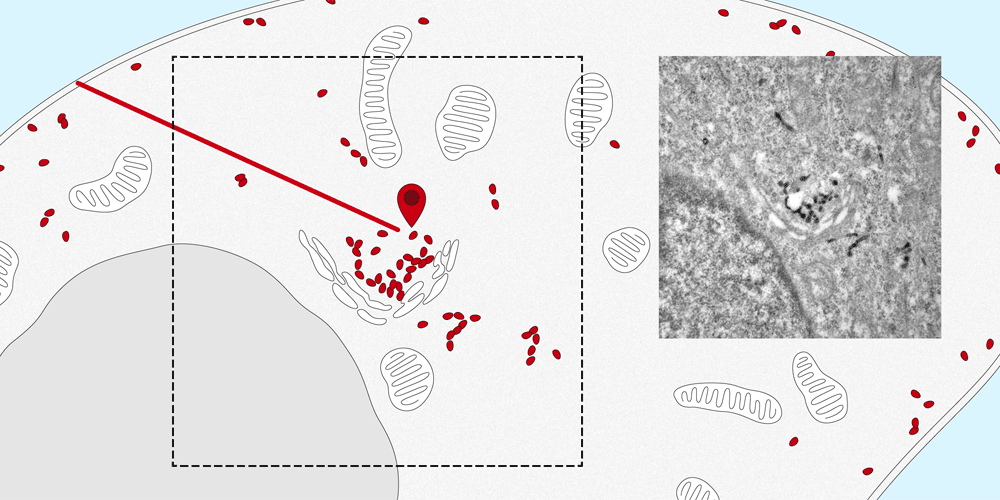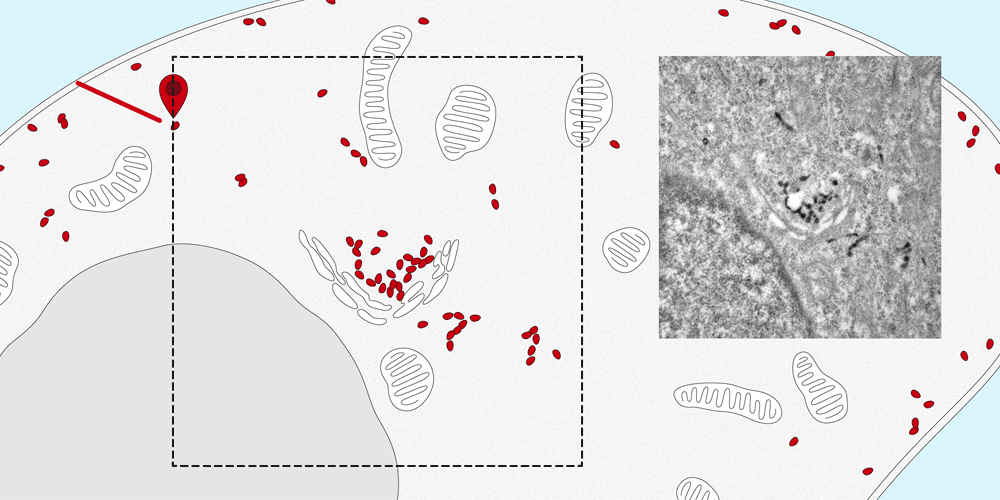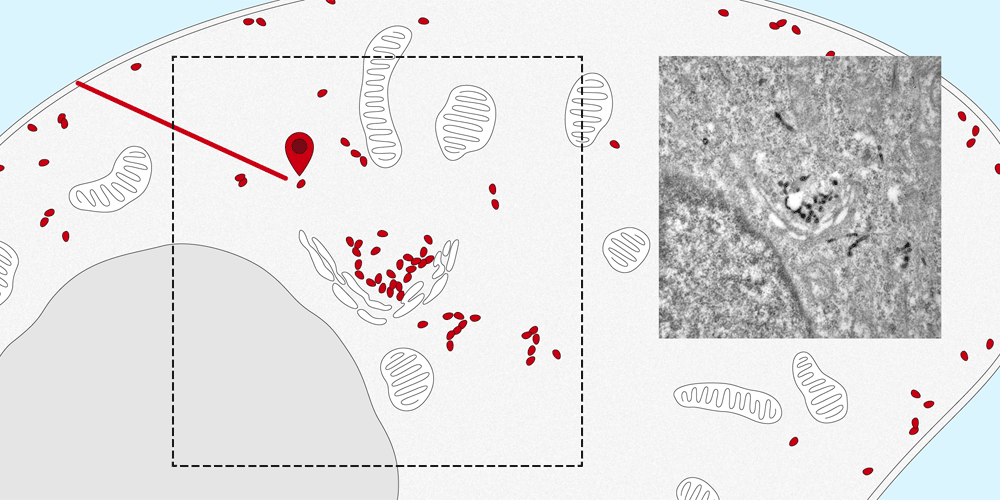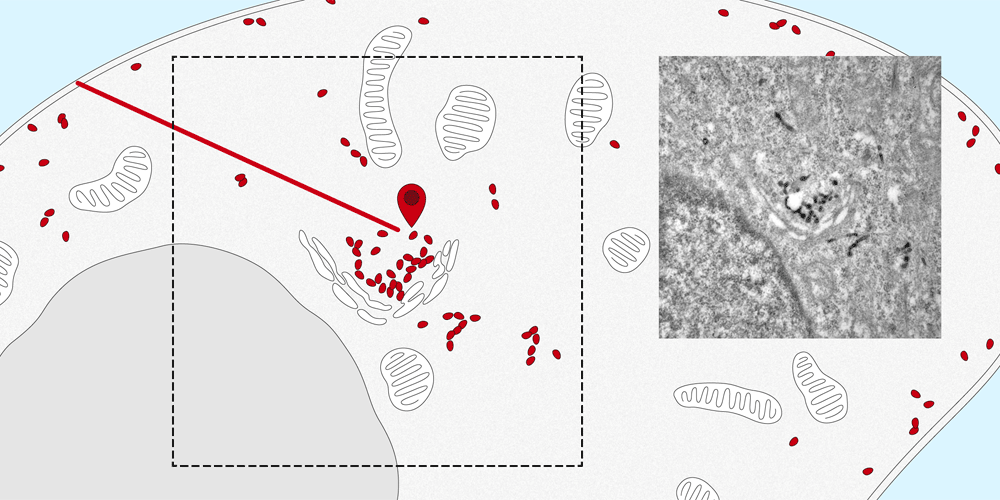
Membrane proteins are a basic component of each individual cell of the human body and play a vital role in the cell’s structure, metabolism and transport. They ensure that many substances, such as hormones and other proteins, are transported from the cell surface into the cell or carried out again.
It is quite easy to determine the distribution of membrane proteins in the cell. Tracking the paths that they take within the cell and identifying their intended destination is considerably more difficult. The new nanobody tool, developed by the Spiess research group at the Biozentrum, University of Basel, makes it possible to observe the movement of proteins into and out of the cell. In the future, this quantitative method can also be applied to elucidate the molecular transport mechanisms inside the cells.
Tiny antibodies as nanosensors
The researchers used so-called nanobodies, tiny antibody fragments. These consist solely of a single protein chain and so, in contrast to antibodies that are composed of four proteins, they have the advantage to be only about one-tenth of the size, very compact and stable. “Nanobodies were originally obtained from camels and lamas. We altered the nanobodies, so that we could produce them with the help of bacteria and use them as nanosensors”, says Professor Martin Spiess.
Nano tags enable live tracking
The nanobodies can be genetically altered to fluoresce. “We attach them like a tag to the targeted protein, where they remain fastened no matter which path the protein takes to enter the cell,” explains Dominik Buser, a postdoc in Martin Spiess’s lab and the first author of the study. Using a microscope, the path of entry and distribution of surface proteins can be observed in living cells.
“The nanosensor with its fluorescent dye makes the exact movements of the proteins visible. This enables us to follow the natural pathways taken by the proteins into the cell, as well as the speed of transport within the cell.” Furthermore, the researchers altered the nanobodies, in a way that the proteins could be localized in the cell by the electron microscope. In the future, the research team plans to apply this new method to track and trace various proteins and to more closely study their transport pathways.
Original article:
Dominik Buser, Kai Schleicher, Cristina Prescianotto-Baschong, Martin Spiess. Versatile nanobody-based toolkit to analyze retrograde transport from the cell surface. PNAS 2018; published online 18 June 2018.
Contact: Communications, Heike Sacher