Main Content
Nano-diffraction of biological specimens
The Abrahams group develops new approaches for using diffraction to determine the atomic structures of proteins and other radiation sensitive nano-size samples. We use diffraction rather than imaging, because diffraction patterns can be measured more accurately than images.
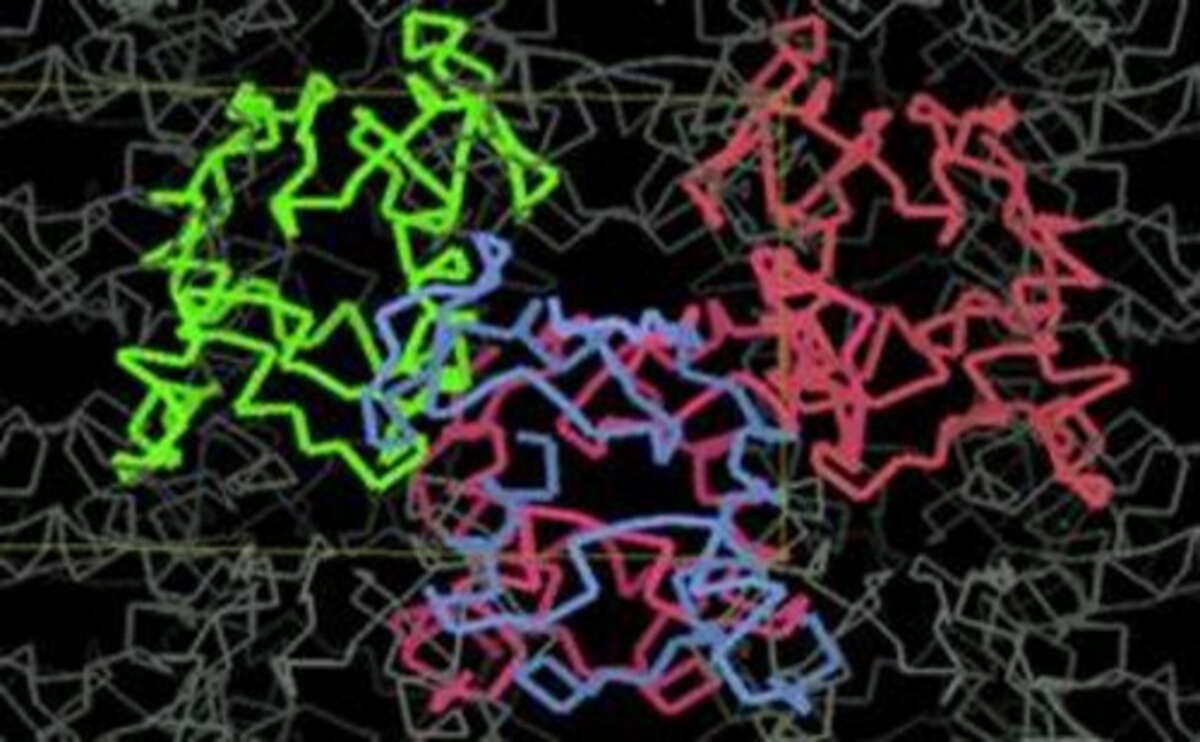
The atomic structure of the protein crystal as deduced from the diffraction patterns.
In first instance, we concentrate our efforts on the structure determination of minute nano-crystals, too small to see even with the best light microscopes. Many proteins can only form such nano-crystals, yet these crystals are too small for standard X-ray diffraction.

A nano-crystal of a protein that is only about 100 nm thick and 200 nm wide. Because this is far too small to see with a light microscope, it is visualised with an electron microscope.
However, nano-crystals are well suited for electron diffraction, because electrons induce far less radiation damage compared to X-rays for the amount of information that their diffraction patterns provide. Electron nano-crystallography of proteins is an emerging technology and many of its aspects need to be developed and improved, while also several fundamental problems remain to be solved.

Three-dimensional electron diffraction of this protein crystal provides atomic information on its structure.
To this aim, we develop novel data collection strategies, implement novel detectors for accurately and sensitively measuring electron diffraction data, we develop novel algorithms for analysing this data and (often in collaboration with others) we investigate new, unkown structures of proteins and other samples. The Abrahams group is located at C-CINA and at the Laboratory of Biomolecular Research at the Paul Scherrer Institute.


