Main Content
Skeletal muscle aging and neuromuscular diseases
Understanding the molecular mechanisms that maintain skeletal muscle structure and function is crucial for developing novel treatment strategies to combat muscle aging and neuromuscular disorders.
Muscle aging
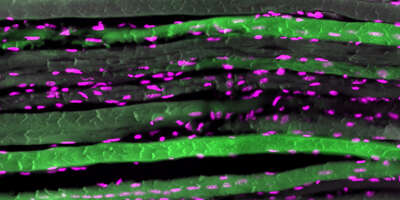
Sarcopenia, the loss of muscle mass and function at old age, impinges on health span and is the major contributor to frailty in the elderly population of industrialized countries. By studying sarcopenia in normal and genetically modified mice, we aim to understand the underlying mechanisms that drive muscle aging.
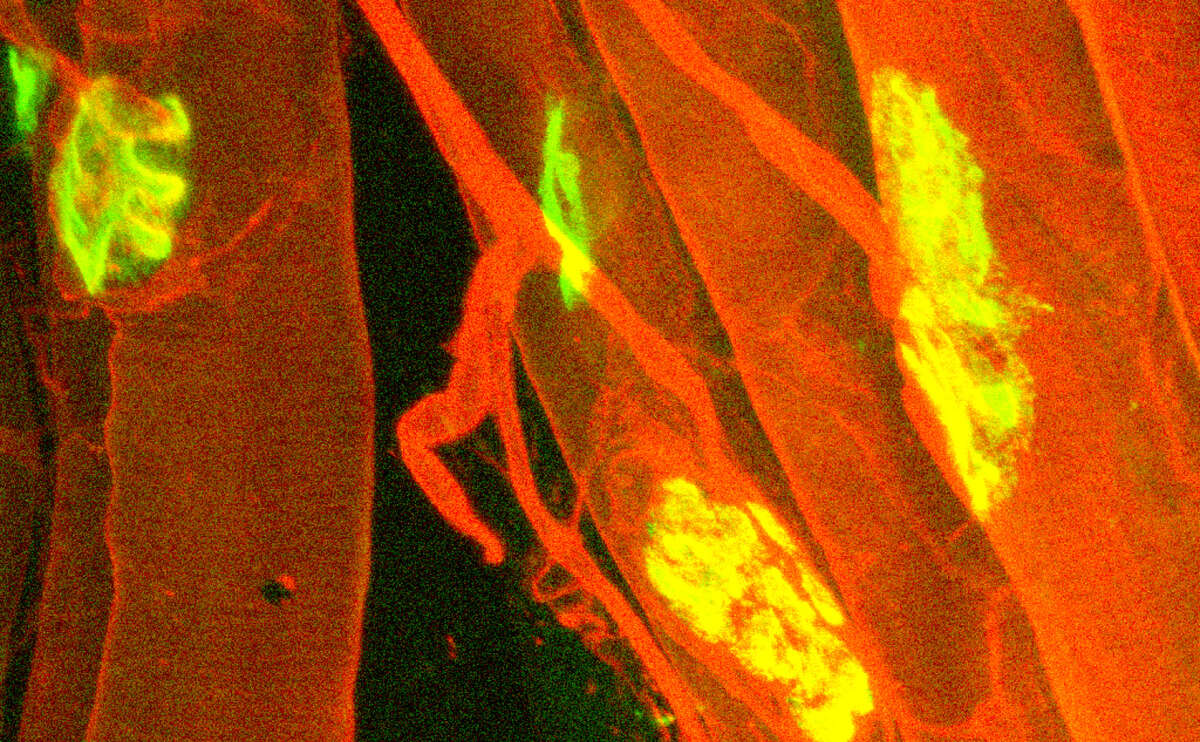
The neuromuscular junction, the site where motor neurons control the contraction of muscle fibers, is a focal point of age-related muscle changes and its stability is paramount to healthy muscle aging. Using the latest techniques, we are now defining the spatial and temporal regulatory networks involved in muscle aging. Using the latest advances in somatic delivery of components required for the CRISPR/Cas9 system, we aim to uncover the significance of the identified regulatory networks on neuromuscular function.
Neuromuscular diseases
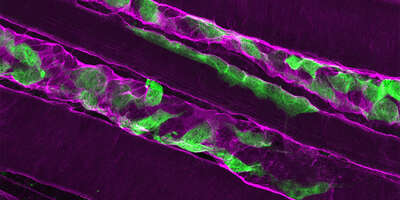
To maintain integrity during forceful contractions, muscle fibers are anchored to an extracellular scaffold of structural proteins. Mutations leading to a loss of key structural proteins are the most common cause of muscular dystrophies. These neuromuscular diseases result in muscle weakness, loss of mobility and even death. Restoring the structural stability of skeletal muscle is the key to treating these diseases.
Recently, we developed a novel strategy to counteract the loss of the structural protein laminin-α2 in a mouse model of this severe, early-onset muscular dystrophy. Using two linker proteins, we were able to compensate the loss of laminin-α2 and stabilize the muscle fibers. We currently investigate the overall potential of these linkers to restore muscle function. Most importantly, by making use of recent advances in gene therapy technology we aim to further develop this strategy with the ultimate goal of developing a therapy for patients.